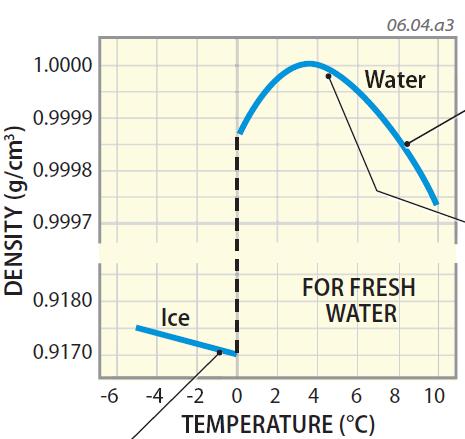
On the other hand, the kinematic viscosity tells about the speed the fluid reaches when a particular force is applied to the fluid. When choosing between the two viscosities, it is worth noting that dynamic viscosity tells us about the force required to move the fluid at a certain speed. The Roman numerals indicate various ice phases.The larger the force or stress needed to move the plate, the more viscous the fluid is. Water at 20☌ can exist in various forms, including freshwater. Definitions, online calculator and figures and tables with water properties like density, specific weight and thermal expansion coefficient of liquid water at temperatures ranging 0 to 360C (32 to 680F). At this temperature, water has a density of approximately 998.2 kg/m³, a dynamic viscosity of approximately 1.002 × 103 Pa·s, and a surface tension of approximately 72.8 mN/m. Water - Density, Specific Weight and Thermal Expansion Coefficients. ‡ Ice XI triple point is theoretical and has never been obtained Phase diagram Log-lin pressure–temperature phase diagram of water. Water at 20☌ is in a liquid state and behaves according to its physical and chemical properties. Table of various forms of ice Properties of various forms of ice Source: CRC Handbook of Chemistry and Physics. Melting point of ice at various pressures ĭata obtained from CRC Handbook of Chemistry and Physics 44th ed., p. 2390 Pure water has a density of 0.99823 grams/cubic centimeter at 1 atm pressure and a temperature of 20 degrees Celsius (68 degrees Fahrenheit). The sixth column is the density of the vapor. The fifth column is the work PΔ V done by each gram of liquid that changes to vapor. The fourth column is the heat of vaporization of each gram of liquid that changes to vapor. The third column is the heat content of each gram of the liquid phase relative to water at 0 ☌. Pressure of the equilibrium is given in the second column in k Pa. On the one hand, the water molecules move faster, on the other hand, in water starts forming clusters. Where P is equilibrium vapor pressure in k Pa, and T is temperature in kelvins.įor T = 273 K to 333 K: A = 7.2326 B = 1750.286 C = 38.1.įor T = 333 K to 423 K: A = 7.0917 B = 1668.21 C = 45.1.ĭata in the table above is given for water–steam equilibria at various temperatures over the entire temperature range at which liquid water can exist. At 4 degrees Celsius in water acts 2 opposing effects, which ‘extend’ water. Vapor pressure formula for steam in equilibrium with liquid water: log 10 P = A − B T − C, The values below 0 ☌ refer to supercooled water.Įlectrical conductivity of highly purified water at saturation pressure Temperature, ☌ Liquid physical properties Temperature dependence of the surface tension of pure water Temperature dependence of the density of ice and water Velocity of sound in water run down the left column for whole degrees then move across for tenths of a degree. Water is the only substance where the maximum density does not occur when solidified. K) at 70 ☌ħ5.58 J/(mol K) and 4.1963 J/(g Fresh water has a maximum density at around 4° Celsius.The pressures are stated in mega-Pascals, where a Pascal is a Newton per square meter, and as a multiple of standard atmospheric pressure. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |




 RSS Feed
RSS Feed
